Which of the Following Substances Can Have Variable Composition
Salt easily dissolves in water but salt water cannot be classified as a substance because its composition can vary. For example water always has the formula H2O sodium chloride common table salt always has the formula NaCl and.
Chemical Composition Chemical Composition Of Substances Chemistry
Mole fraction mass fraction and concentration can be used as composition variables for both solvent and solute just as they are for mixtures in general.

. The correct option is C AIR. A must be an element. The number of known pure substances is less than 100000.
E A heterogeneous mixture has a variable composition throughout. The constituents of air can vary. A pure substance cannot be separated into other kinds of matter by any physical process but a mixture can as they are impure substances because they contain more than one particles.
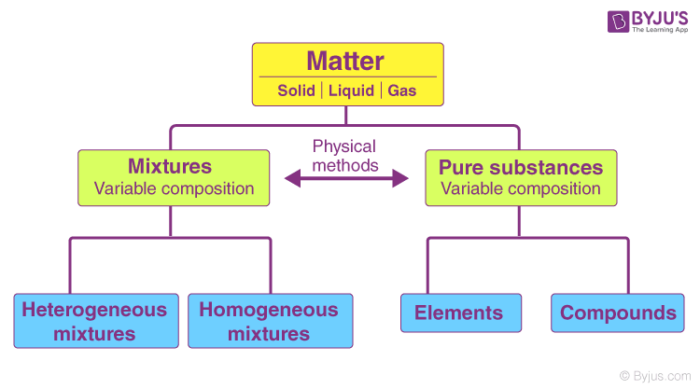
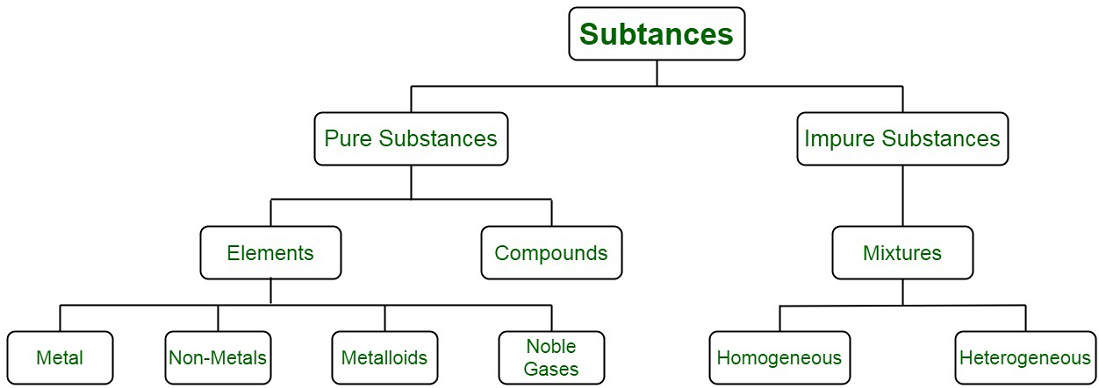
Pure substances are mostly homogeneous in nature containing only one type of atoms or molecules 2. Air is made up of different types of elements which are not chemically combined together therefore air is a mixture. Water is also a pure substance.
A salad with lettuce cheese seeds tomatoes broccoli and other vegetables is an example of a heterogeneous mixture. Two substances A and B were made to react to form a third substance A2B according to the following reaction. The substances have fixed boiling and melting points 4.
The study that is concerned with composition transformation and characteristics of matter is known as chemistry. Which one of the following statements about mixtures is correct 2 3 The different substances in a mixture are always in a definite proportion to cacho A mixture is made up of elements that are chemically combined It is extremely difficult to separate a mixture into different substances A mixture must contain at least two different substances A mixture is. C Metallic sodium is soft enough to be cut with a knife.
I am not 100 sure but i believe variable composition is when the composition of a mixture can vary or contain different properties elementscompounds. C must be a pure substance. It is considered a substance because it has a uniform and definite composition.
A The composition of a sample of steel is 98 iron 15 carbon and 05 other elements. In a compound containing copper and sulfur the two elements are. All homogeneous mixtures must contain at least two substances.
A mixture containing copper and sulfur can have a variable composition. Compounds have a definite composition therefore they are pure substances. The Characteristics of the Mixture are as Follows-.
Given statement Heterogeneous mixtures but not homogeneous mixtures can have a variable composition is true or false has to be classified. Correct option is E A homogeneous mixture has the same uniform appearance and composition throughout. Two substances A and B were made to react to form a third substance A2B according to the following reaction 2A B A2 B.
In a compound containing copper and sulfur physical methods can be used to separate the substances present. B Compounds are composed of two or more elements in fixed proportions. D Compounds cannot be decomposed into simpler substances by chemical means.
These substances mainly have a constant or uniform composition throughout 3. Heterogeneous mixtures but not homogeneous mixtures can have a variable composition. An example of variable composition.
Ordinary table salt is called sodium chloride. I The product A2B shows the properties of substances A and B. Pure substances cannot be separated into other kinds of matter by physical means.
D more than one correct response. E no correct response. A mixture containing copper and sulfur can have a variable composition TRUE Shannon b In a mixture of copper and sulfur the two elements maintain their individual properties TRUE.
The proportions in which the substances are dissolved is variable. I The product A 2 B shows the properties of substances A and B. C A solution is a homogeneous mixture.
The separations of the substances from the mixture can easily be done as it is just a mechanical blending process. B could be a compound. C Pure substances cannot have a variable composition.
Ii The product will always have a fixed composition. In a mixture of copper and sulfur the two elements maintain their individual properties. Which of the following substances can have variable composition.
Which of the following statements concerning this reaction are incorrect. B Zinc dissolves in hydrochloric acid with the evolution of hydrogen gas. E no correct response.
The substance does not share any chemical bonding while in a mixture. T In a compound containing copper and sulfur physical methods can be used to separate the substances present. NaClaq solution and milk is a homogeneous.
Alcohol and water would be a real homogeneous mixture gin or vodka can be kept in a bottle forever without separating and the percentage of alcohol can be highly variable. 127 d A substance which cannot be separated into two or more substances using physical means. A Different mixtures of the same substances can have different compositions.
Ii The product will always have a fixed composition. D more than one correct response. All samples of sodium chloride are chemically identical.
Pure substances do not have fixed composition whereas mixtures may contain the components in any fixed ratio b Pure substances have fixed composition whereas mixtures may contain the components in any ratio and their composition is variable. A heterogeneous mixture consists of visibly different substances or phases. 2A B A 2 B Which of the following statements concerning this reaction are incorrect.
Apr 112022 - Which of the following statements is trueaHomogeneous mixtures can have variable compositionbHomogeneous mixtures fixed compositioncHeterogeneous mixtures have fixed compositiondSalt solution is heterogeneous mixtureCorrect answer. A pure substance usually participates in a chemical reaction to form predictable products. T In a mixture of copper and sulfur the two elements maintain their individual properties.
A pure substance has fixed composition as. A mixture containing copper and sulfur can have a variable composition. A fourth composition variable molality is often used for a solute.

Helpful Chem Flowchart Flow Chart The Moon Is Beautiful Welcome To Night Vale

Pin On Chemistry Classification Of Matter

What Are The Types Of Pure Substances Compounds Elements Videos

Matter Chemical Substance Classisification Chemogenesis Chemistry Lessons Classroom Management Techniques Elements Compounds And Mixtures

Here S The Poppy I Ve Been Wanting To Show You A Few Of Them Have Sprung Up In The Front Garden For The First Time This Year And T Front Garden Poppies

Classification Of Matter Blank Flowchart Thursday November 15 2018 Therapy Worksheets Printable Worksheets Worksheet Template

Pdf Chapter 2 Matter And Change 11 Section 2 1 Properties Of Matter Pages 39 42 Aidan Dudley Academia Edu
What Is Pure Substance Definition Examples Difference Between Pure Substance Mixture

Pure And Impure Substances Geeksforgeeks

The Science Of Coffee Black River Roasters Coffee Roasting Coffee Infographic Coffee Roastery

Pure Substances And Mixtures Elements Compounds Classification Of Matter Chemistry Examples Youtube

Pure And Impure Substances Geeksforgeeks

Pin By Ramesh Thiyagarajan On Science Medical School Studying Medical School Organization Biochemistry

4 Natural Skincare Ingredients That Are Way More Effective Than Synthetic Chemicals Natural Skin Care Ingredients Natural Skin Care Excessive Dry Skin

9th Class Science Notes In English Chapter 2 Is Matter Around Us Pure Gyan Study Point Science Notes Matter Science Physics Notes

Matter Chemical Substance Classisification Chemogenesis Matter Teaching Pure Products


Comments
Post a Comment